Redefining Protecting Groups through Unconventional Reactivity of Nms-amides
Institute of Organic Chemistry, University of Vienna, Währinger Straße 38, 1090 Vienna, Austria
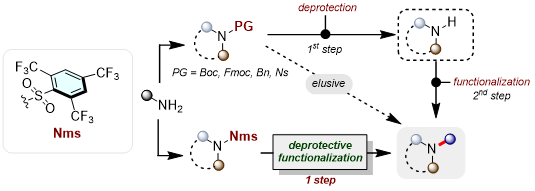
The nature of protecting group chemistry necessitates a deprotection step to restore the initially blocked functionality prior to further transformation. As this aspect of protecting group manipulation inevitably adds to the step count of any synthetic sequence, the development of methods enabling simultaneous deprotection and functionalization – beyond a “one pot, two-step deprotection followed by functionalization” – is appealing.1 Recently, our group has established a new sulfonyl protecting group which we termed Nms (Nonafluoromesitylenesulfonyl) exhibiting broader and complementary applicability than benchmark sulfonamides (Ts, Ns, Cs).2 Herein, we describe how the newly introduced Nms-amides enabled the development of the concept of “Deprotective functionalization”,3 as we navigate their direct conversion to valuable nitrogen-containing molecules.4,5 Mechanistic and case studies further substantiate and help rationalize the exquisite reactivity of Nms-amides.
Figure 1. General concept of deprotective functionalization of Nms-amides.
References
1. Young, I. S.; Baran, P. S. Nat. Chem. 2009, 1, 193–205.2. Spieß, P.; Sirvent, A.; Tiefenbrunner, I.; Sargueil, J.; Fernandes, A. J.; Arroyo-Bondía, A.; Meyrelles, R.; Just, D.; Prado-Roller, A.; Shaaban, S.; Kaiser, D.; Maulide, N. Chem. Eur. J. 2023, 29, e202301312. 2. Spieß, P.; Sirvent, A.; Tiefenbrunner, I.; Sargueil, J.; Fernandes, A. J.; Arroyo-Bondía, A.; Meyrelles, R.; Just, D.; Prado-Roller, A.; Shaaban, S.; Kaiser, D.; Maulide, N. Chem. Eur. J. 2023, 29, e202301312. 3. Tiefenbrunner, I.; Shaaban, S.; Maulide, N. Chem. Eur. J. 2026, 32, e03271. 4. Spieß, P.; Brześkiewicz, J.; Meyrelles, R.; Just, D.; Maulide, N. Carboxylic Acids. Angew. Chemie Int. Ed. 2024, 63, e202318304. 5. Spieß, P.; Brześkiewicz, J.; Maulide, N Chem. Sci. 2024, 15, 15799–15803.
